
Section 204: Impacts on your Traceability Program
Daria Van De Grift
Section 204 is a part of the greater Food Safety Modernization Act (FSMA) regulation passed in 2010 and signed by President Barack Obama in 2011. Section 204 focuses on the Food Traceability list (FTL) was finalized and released in November 2022. The FDA requires compliance in three years, which means by January 2026 all companies that interact with foods on the FTL must meet the standards set forth by 204. Three years may seem like a lot of time but the best advice from industry professionals is not to wait.
Food Traceability List
The FTL was determined by a risk-based model and is to be reevaluated every 5 years. Therefore, if a food is not on this list now it may be in the future. The risk-based model used the following factors ¹;
-
Frequency of outbreaks and occurrences of illnesses
-
Severity of illness
-
Likelihood of contamination
-
The potential for pathogen growth, with consideration of shelf life
-
Manufacturing process contamination probability and industry-wide intervention
-
Consumption rate and the amount consumed
-
Cost of illness

Because of the criteria, food on this list may be added or removed depending on if it fits the criteria.
There are exemptions for foods on this list and the FDA has a tool to help you decide if you do fall into this category. To see if you are exempt please follow the link to take a short quiz from the FDA
A few ways a food on the FTL could be exempt is if it is processed downstream in a way that kills the target pathogen by a 5-log reduction. Another way a product could be exempt is if it is already exempt from other FSMA rules like the produce rule. Lastly, food may be exempt through written assurance that a processor will apply a kill step.
Additional Traceability
There is some new vocabulary that applies to this rule. The first is a Critical Tracking Event (CTE) which includes Growing, Transformation, Creation, and Transportation². These events trigger a Key Data Element (KDE) which is the documentation that captures the food safety or quality parameters at that event³.
An additional new phrase being used is the Traceability Lot Code. This is a special lot code that is assigned to the product that is on the FTL from the first time it starts from the farm to the time it reaches the customer.
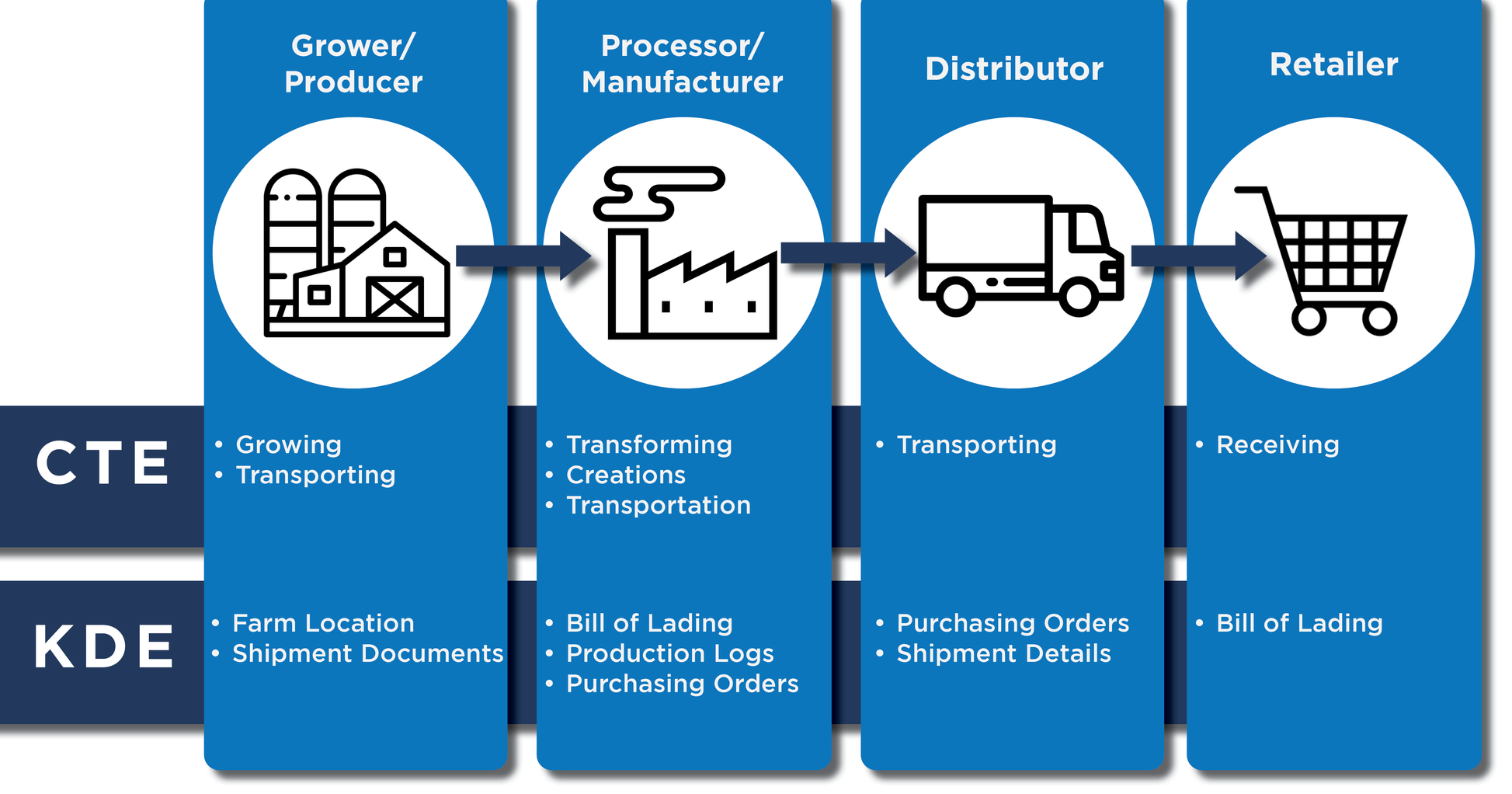
All of the documentation that is now required to be passed from farm to retailer means that you will need a fast, efficient, and simple way to obtain and send those documents. The best way to manage this is through electronic document management software like Document Compliance Network (DCN).
DCN makes receiving and sending documents along the supplier chain super easy. Suppliers are able to upload documents straight from their email without needing to create and account, log in, or do anything hard. Then you can connect your documents to your customers, as well as supplier documents to them as well. DCN also automatically sends out a notification when documents are missing, expiring, or expired. Using an electronic document management system also helps to comply with the 24-hour requirement from the FDA to provide all traceability documents in the event of a recall.
Tips for implementation
The biggest tip is to not wait until the last minute to start! You have 3 years to comply but you need to allow time for you to work with your supply chain to start testing the flow of documents from them to you. Then you need to TEST your system! Once you have made the changes to your programs you need to test them based on how the FDA will audit you. Because 204 involves the retailers that your product is sold at they may try and test the system before the 3-year mark as well.
The next piece of advice is to cross-train your teams on how the process works. You do not want to be the only one that knows how to use the software or complete the recall procedure. It also helps to have several people on the team so that if one is out you are not scrambling to meet the requirements.
To schedule a demo with Document Compliance Network, click here.
Sources
- 18 Views
- 0 Comments


